| Home |  | Patient Safety |
Synjectos®
using its in-house development process, confirming that the material will fulfill all specifications in terms of function, purity and bio-burden and will apply for registration in Indonesia and the rest of SE Asian region.
using its in-house development process, confirming that the material will fulfill all specifications in terms of function, purity and bio-burden and will apply for registration in Indonesia and the rest of SE Asian region.
Our
in-house experts will also supervise the production process of
"Synjectos" in the SE Asian region. They also give training and
education too personal, distributors, surgeons and surgeons staff.
Regarding
the patient safety and performance of “Synjectos®”, said material will end up
into a compressive strength of > 40 MPa after 40 hours of incubation, which
exceeds the strength of human trabecular bone which is reported to be up to 30
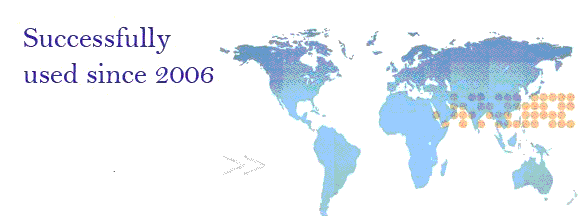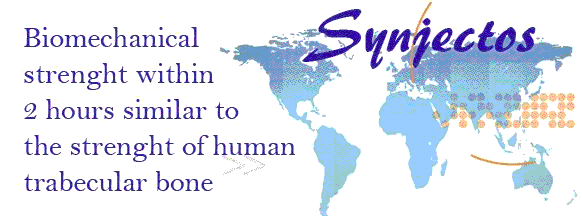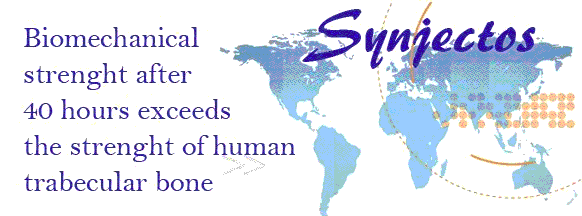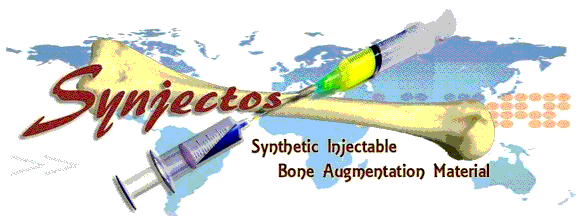
MPa). Even 2 hours after incubation of specimen prepared due to ISO EN 5833 for
compressive strength testings are reaching strength of more than 20 MPa.
Considering that bone voids which are augmented with autologous cancellous bone
graft or allograft material can reach a strength of about 5 MPa only, it is
obvious that the bio-mechanical strength of the material is sufficient to act as
a cancellous bone graft or bone filler material.
Given the fact that the European
similar product has been successfully used in more than 20.000 patients and the
composition of “Synjectos®” is almost identical, made from material with the
same analytical grade purity and reaching the same magnitude of strength, it
can be concluded that this material is safe.


